In this week’s blog post we will take a deeper look at one of the new models from the last version (2022.1), and what theory it brings to the table. The new liquid-solid-gas mixture model for Lagrangian particles. The model is referred to as “REA Spray drying Evaporation” (Reaction Engineering Approach). It has its main application in simulating milk droplets in spray drying or the process of drying wet solids (CPI).
The video below shows the process of milk droplets drying in a counter flow spray dryer.
What is the REA-model?
The general REA model is a way to model drying kinetics by applying chemical reaction engineering principles. It was created in 1996 and seen in a publication in 1997. In REA, evaporation is modeled as zero-order kinetics with activation energy. The condensation on the other hand is treated as a first order wetting reaction with respect to drying air solvent vapour concentration without activation energy. Zero-order kinetic means that the rate of reaction is not a function of concentration. While first order kinetics depends linearly on only one reactants concentration.
The model offers two distinct sub models, the L-REA (lumped REA) and S-REA (Spatial REA), where the S-REA is basically an extension of the L-REA. In the lumped REA, we model global drying rate with the assumption that the activation energy is constant. With Spatial REA we introduce an expression for the activation energy, and the possibility to expand the capabilities of REA to e.g. intermittent drying under time varying temperature, humidity, and infra-red heating etc. Essentially, we can couple the global L-REA model to a system of equations of conservation to yield the spatial S-REA-model. Without modifications the L-REA approach can model convective drying of the mixture of polymer solutions accurately. Since the model is partly empirical, and require measurement input, the L-REA parameters obtained can also be used for the S-rea approach.
It has its basis in, and can be compared to Fick’s law of diffusion. Fick’s law is one of the most well-known concepts of mass transfer in literature. Using the same mathematics as in Fourier’s law of heat conduction or Ohm’s law of electrical conduction, diffusion can be expressed as:

This formulation represents one dimensional diffusion. But, due to the complexity of the process of drying a porous material an “effective diffusivity” is calculated to capture more effects that just the pure diffusion process.
The advantage of REA generally is that it offers the advantage of drying to be expressed in terms of simple ordinary differential equations (a function containing derivative expressions, only based on one unknown) and this removes the complication of using partial differential equations (where there are derivative expressions of several unknowns).
Under the hood
The REA-formulation used in Simcenter STAR-CCM+ can be seen in picture below. What the model does is controlling the moisture removal rate by correlating the instantaneous surface vapour density with its saturated value.

During the initial stage of the drying, the particle is entirely coated with liquid and by equilibrium assumption, the vapor density is equal to the saturated value and the rate reduction factor is 1. This leads to the formulation for evaporation of a pure liquid droplet. As, the drying continues, the rate reduction factor is progressively reduced, due to the increase of solid fraction on the particle surface that limits moisture removal. The rate reduction factor (the “fraction” for enabling drying) is modeled according this the below picture.
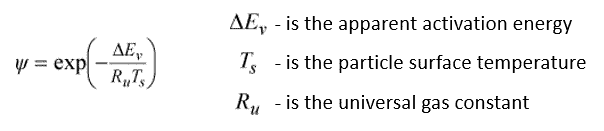
Under the assumption of a small Biot number, the surface temperature is approximated with the particle temperature. The Biot number describes the relationship between the thermal resistance inside a body and at the surface of a body, determining if there is a strong gradient inside the body [Dimensionless numbers – VOLUPE Software]. This assumes that the evaporation is an activation process, meaning that the evaporation need to overcome an energy barrier, e.g. as a result of the formation of crust on the droplet surface. The apparent activation energy represents a “resistance” to extracting moisture, regardless of drying stage.
Further expansion of the capabilities of the model for varying drying gas conditions, the apparent activation energy is normalized with its equilibrium value of the drying gas.
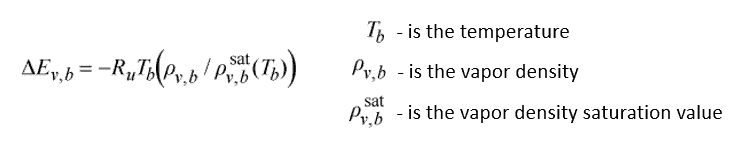
The normalization makes it so that the activation energy is only a function of the deviation of the moisture content on the particle surface from its equilibrium value in the drying gas. What is then used is a model for the normalized activation energy. The term X_eq in the model for normalized activation energy is the equilibrium moisture content and is in turn a function of water activity. The water activity (a_w) related to water in food material, is defined as the ratio of partial vapor pressure of water in the sample to the partial vapor pressure of pure water at the same temperature. And this in turn, is solved by using another model called the GAB model (Guggenheim-Anderson-de Boer model).

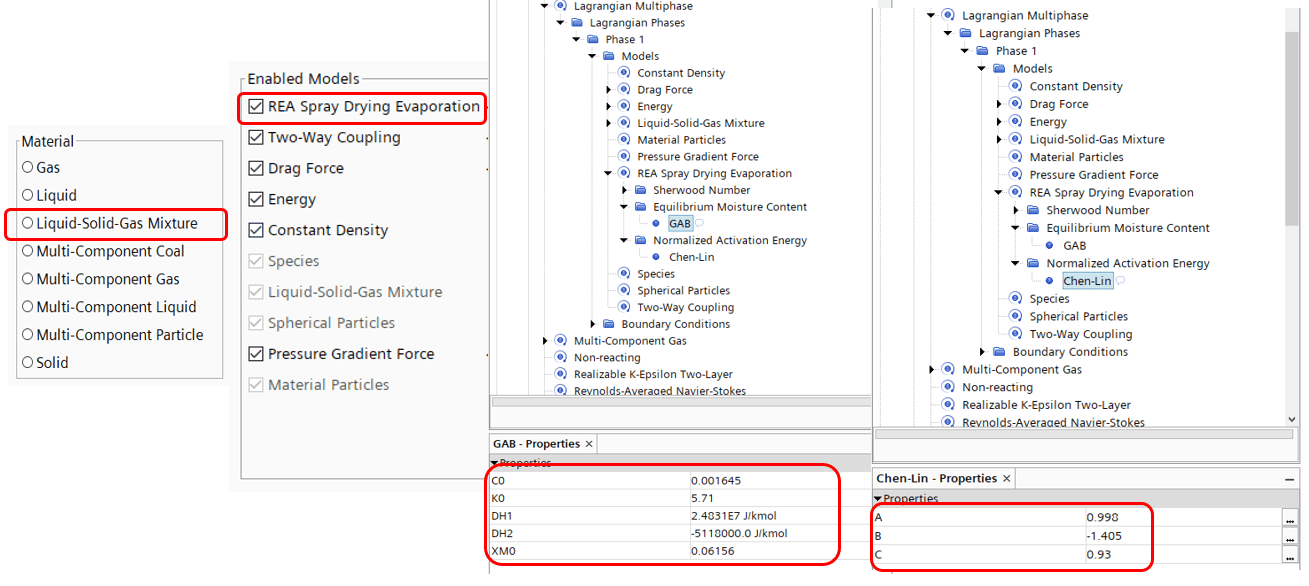
Note that both of these models are packed with constants (a, b and c for Chen-Li and C_0, K_0 and X_mo for GAB) and the values for these constants can be found in the documentation for Simcenter STAR-CCM+ under “Particle Heat and Mass Transfer” in the theory section for the Lagrangian approach. It should be noted that the default values, pre-selected when using the model pertains to a 20 wt% skim milk solution. Correlations for different materials and initial compositions can be found from literature and implemented with user-defined field functions. (An even more extensive description of the models and equations can be found under the documentation section previously mentioned)
In Simcenter STAR-CCM+
Now, we have looked under the hood for this new model, but what selections do we do in the software to use this model? It is quite straight forward, when creating your Lagrangian phase you select the new node of “Liquid-Solid-Gas Mixture”. In addition you enable the REA-Spray drying evaporation model. Note that there is also a model for Multiphase mixture evaporation that can be selected instead of the REA model. That model works exactly like the model for Multi-component Droplet evaporation. Further you can find all the constants relating to the previously mentioned models in the properties for each model. And from this point you need to define the components in your mixture, and here you can now include a solid, something that was not possible before. From here on the procedure is the same as in earlier versions.
I hope this has been useful and that this has given some insight into the new model available in STAR. Do not hesitate to reach out at support@volupe.com if you have any questions.
Author

Robin Victor
+46731473121
support@volupe.com